AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
(e) Burning biofuels contributes less to climate change than burning fossil fuels. Explain why increasing the use of biofuels may cause food shortages. ġ0 ☌ 25 ☌ 350 ☌ (d) Ethanol produced by fermentation is often added to petrol to create a biofuel. Draw a ring around the temperature that is most suitable for this process. Ethanol can also be produced by the fermentation of sugars from plants. 
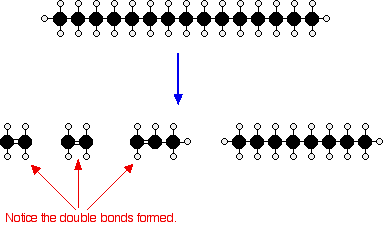
Use image Add the missing bonds to complete the displayed structures in Figure 1. 
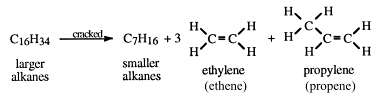
Use image Give the formula for this product and explain why the chemical formula of this compound is different to the others You should include the name of the family of compounds. (b) State whether C12H26 has a higher melting point than either C4H10 or C6H12 and explain why. C12H26 → C4H10 + C6H12 + C2H4 Give two conditions necessary to crack large alkane molecules. The equation shows the cracking of dodecane. (a) Long chain alkanes are routinely cracked to produce more useful molecules. Most hydrocarbons extracted from crude oil are alkanes. (d) Naphtha is a fraction obtained from crude oil with the formula C7H16. Write down the balanced equation for the combustion of propane, C3H8. Use image (a) Write down the molecular formula of the small molecule from which polythene is made. (d) Use the graph to determine the boiling point of butane, showing clearly how you arrived at your answer.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
